Improvements in global health since the introduction of antimicrobials are being threatened by microorganisms that have become resistant to a wide range of antibiotics, antivirals, antiseptics, and antifungals.
An antimicrobial is any natural, synthetic, or combined substance that kills or stops the growth of microorganisms such as bacteria, fungi, protozoa, and viruses. Antimicrobial substances include drugs such as antibiotics, antivirals, and antifungals, and chemicals such as antiseptics, disinfectants, and sterilizers.[1]
The introduction of the first antibiotic, penicillin, in 1941 transformed the treatment and management of many infectious diseases. Unfortunately, many of today’s human diseases such as bloodstream infection, HIV/AIDS, malaria, pneumonia, tuberculosis (TB), and urinary tract infections (UTIs) are now resistant to a wide range of antimicrobials.[2] According to a 2019 analysis on the global burden of bacterial antimicrobial resistance (AMR), an estimated 4.96 million deaths that year were associated with AMR, including 1.27 million deaths directly attributable to bacterial AMR. This was significantly more than deaths attributable to HIV (680,000) and malaria (627,000) combined.[3] The World Health Organization (WHO) has declared antimicrobial resistance (AMR) to be one of the top 10 global public health threats now facing humanity.[4]
What is AMR?
Antimicrobial resistance is the inherited or acquired ability of microorganisms to survive or proliferate at concentrations in the presence of an antimicrobial that would otherwise kill or inhibit them.[5]
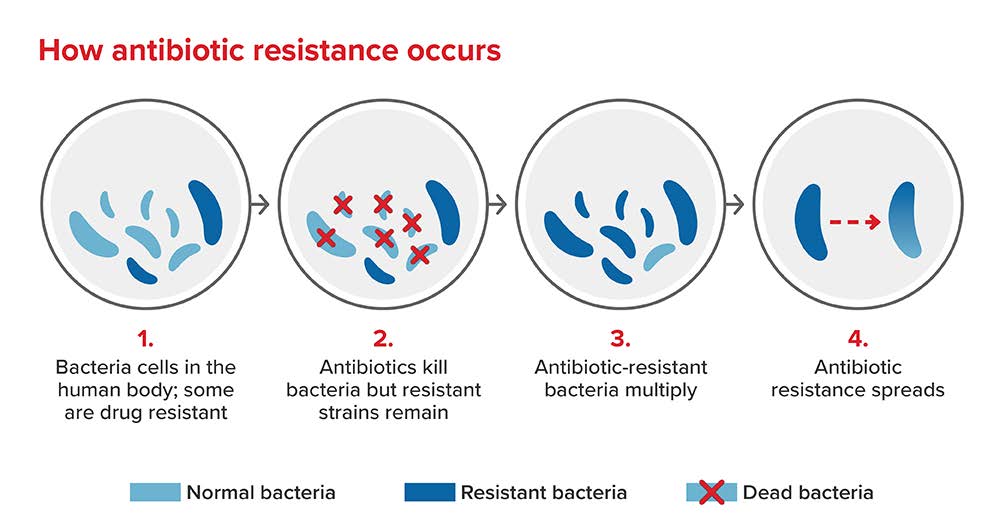
Bacterial AMR occurs when changes in a bacterial strain cause the pharmaceutical(s) designed to treat an infection to become less effective. These microorganisms can develop resistance following contact with other resistant microorganisms or from overuse of antimicrobials and can adapt and grow even when treated with antibiotics. Once they develop resistance, they can multiply and become resistant to multiple agents, and are referred to as “super-bugs.” Resistance can also be shared among bacteria through genetic alterations. The exchange of genetic material between different bacteria can affect antibiotic treatment for a wide range of infections, making them untreatable. Drug-resistant bacteria are then spread and circulated between humans, animals, and the environment.[1] [6]
Figure 1:
What are the main causes of AMR?
The key causes of AMR are the overuse or inappropriate use of antibiotics in humans, animals, food production, and agriculture, as well as poor waste water management and hygiene, particularly in lower-income countries. Each year in the U.S., out of 40 million people treated with antibiotics for respiratory infections, 27 million receive them unnecessarily, while 13 million who need them do not receive them.[7]
Evidence suggests that the release of antimicrobials into the environment, when combined with natural and resistant bacteria, is leading to the evolution of more resistant strains of bacteria.[1] Higher global temperatures have also been associated with an increase in the spread of bacteria, viruses, parasites, and fungi, causing an increase in AMR infections.[5][6]
Bacteria
Many infections are caused by five primary antimicrobial-resistant bacteria: Escherichia coli (E. coli), Klebsiella pneumoniae (K. pneumoniae), Enterococcus faecium, Pseudomonas aeruginosa, and Staphylococcus aureus (MRSA). In high-income regions, nearly half of fatal AMR deaths have been linked to MRSA and E. coli. Resistance to first-line antibiotics accounted for more than 70% of deaths due to AMR.[8] Patients with MRSA infections resistant to treatment are 64% more likely to die than patients who respond to antibiotic treatment. For E. coli, rates of resistance to the antibiotic ciprofloxacin range from 8.4% to 92.9%, and for K. pneumoniae range from 4.1% to 79.4% globally. K. pneumoniae is a major cause of hospital-acquired infections, while Acinetobacter baumannii is often detected in patients with weakened immune systems and is emerging as an additional bacterium increasingly resistant to antibiotics. [9]
Risks
AMR poses a serious threat to human health. It has been estimated that as many as 10 million people could die each year from AMR by the year 2050, a number equivalent to the 2020 death toll from cancer. Surgical operations and cancer chemotherapy will become less safe due to antibiotics becoming less effective, or even ineffective, in protecting against infection. Once-routine operations could also become fatal by 2050 due to resistant bacteria.[5][8]
Increasing antibiotic persistence, that is, the ability of a bacterial population to survive exposure to an antibiotic to which the bacteria are susceptible, is resulting in the use of second- and third-line antibiotics, which are generally considered a line of last resort. These antibiotics can cause serious side effects such as organ failure, and many patients using them ultimately require extensive long-term care.[10]
Emerging drug-resistant HIV strains pose another significant concern, with antiretroviral (ARV) drugs used to treat HIV at risk of becoming ineffective. Levels of HIV infections resistant to ARV non-nucleoside reverse-transcriptase inhibitors (NNRTIs) among adults receiving first-line therapy has exceeded 10%, and more than 50% of newly diagnosed HIV infections among infants in sub-Saharan Africa were found to be resistant to NNRTIs.[9]
How has AMR been affected by the COVID-19 pandemic?
Although numerous people have been admitted to hospitals for SARS-CoV-2, the virus responsible for COVID-19, there have also been large reductions in hospital admissions since the start of the pandemic for causes other than COVID-19, due to cancellations of elective procedures and reduced referrals for ill health. This subsequently led to reduced usage of antibiotics but increased usage of antifungals in hospital settings, due to the number of individuals who required intensive care treatment. While resistant infections and deaths were estimated to have dropped by 15% and 17% respectively in 2020 compared to 2019, the overall burden of AMR increased by 4.5% between 2016 and 2020.[11]
Tackling AMR
Measures to combat AMR include developing new antimicrobial drugs to treat infections, using vaccines to prevent infectious disease, employing better infection surveillance, reducing the use of unnecessary antibiotics, increasing the use of appropriate rapid diagnostic tests, and improving hygiene and water sanitation.[7]
Although there is increased awareness of the need to develop new classes of antibiotics, no major new class of antibiotic has been discovered since 1987.[6]
The development of new antimicrobials has been slow, given the greater focus on developing treatments for non-infectious diseases such as cancer, cardiovascular disorders, and dementia. It has also proven difficult to find new preparations that are effective in treating bacterial infection. As of December 2020, 43 antibiotics were in development, of which 13 were in Phase 3 clinical trials (approximately 60% of drugs in Phase 3 clinical trials are approved).[12]
Vaccine development is vital to preventing further increases in AMR. Vaccines not only save lives but also reduce levels of AMR. The use of pneumococcal vaccines, for example, has been shown to lead to a 64% reduction in antibiotic resistant infections in children and a 45% reduction in adults over 65 years of age.[7]
The extended use of laboratory testing and new diagnostic tools are crucial to identify specific infections and stop the inappropriate use of antibiotics. C-reactive protein (CRP) testing, for one, can provide indication of infection and has reduced antibiotic prescribing for respiratory tract infections by 25%.[4]
How is AMR likely to affect the insurance industry?
The long-term consequences of AMR may be significant, resulting in increased infection and illness, longer duration of infection, increased hospital admissions and length of hospital stay, and reduced protection from infection during surgery or chemotherapy. Already, at least 1.7 million adults in the U.S. develop sepsis each year from infection.[13] The cost of medical care will therefore increase as more people become resistant to antimicrobials and require prolonged treatment and hospital admission. All these outcomes may eventually lead to increased claims and insurance costs. Over time, mortality rates may also rise as fewer people respond to existing treatments and succumb to once easily treated infections.[6]
Summary
AMR has long-term environmental, health, and financial implications. It is a major threat to human health, but measures are being put in place to address the crisis. These include investment in developing new antimicrobials and alternative treatments, increased vaccination against infection, and a reduction in the use of unnecessary antibiotics in both humans and animals. While these current measures will better prepare populations against antimicrobial resistance that continues to emerge worldwide, ongoing vigilance in the years ahead will be required to avert a major crisis.