Head and neck cancers (HNCs) are defined as those of the upper aerodigestive tract, which includes the oral cavity, the mucosal lip, the oropharynx, the hypopharynx, the nasopharynx, the larynx, and the salivary glands.
HNCs were the eighth most common cancers worldwide in 2018 and accounted for 3% of all cancer diagnoses and about 1.5% of cancer deaths in the U.S. alone. Up to an estimated 85% of all HNC risk today is due to smoking and other tobacco product use, and alcohol consumption is also an important independent risk factor. Human papillomavirus (HPV) is an emerging risk factor as well, specifically for oropharyngeal cancer, the most common type of HPV-associated cancer, which has patient demographics that differ substantially from those of non-HPV associated HNC cases. Survival rates for HNCs, according to Surveillance, Epidemiology, and End Results (SEER) data, show overall survival for localized cancers range from more than 90% for salivary gland and mucosal lip sites to closer to 60% for laryngeal and hypopharyngeal sites.
Several important changes were made in HNC staging in the AJCC Cancer Staging Manual, Eighth Edition, released by the American Joint Committee on Cancer on January 1, 2018. This article updates the reader on HNC and reviews the staging changes.
Epidemiology and Risk Factors
Head and neck cancers (HNCs) are specifically defined as cancers of the upper aerodigestive tract, which includes the oral cavity, the mucosal lip, the oropharynx, the hypopharynx, the nasopharynx, the larynx, and the salivary glands.1 Globally, HNCs were the eighth most common cancer in 2018, accounting for 3% of all cancer diagnoses and 1.5% of cancer deaths in the U.S. alone.2
More than 90% of all HNCs are squamous cell carcinoma (SCC), arising in the mucosal surfaces lining the aerodigestive tract.3 The oral cavity is the most common site of occurrence.4, 5
HNCs have several risk factors. The most common are:
- Tobacco use. Up to 85% of HNC risk is estimated to be due to smoking and other tobacco product use.6 For heavy smokers, the risk of HNC is five to 25 times higher than it is for nonsmokers.7 As global rates of smoking and tobacco use have declined, so have rates of tobacco-related HNC. This decline, however, has not been uniform. Rates have dropped mostly in North America and Western Europe,8 but remain high in Eastern Europe and throughout Asia – especially in China, India, and Indonesia, where half of the world’s male smokers currently reside.9
Several modes of tobacco use specific to South and Southeast Asia may also be predisposing individuals to oral cancers, due to how they are used and the components with which tobacco is combined.10 Use of betel quid, for example, is common in both regions as well as among populations that emigrated from these areas, and may be responsible for the high prevalence of oral cancer in these populations.11 Betel quid use involves either chewing or placing between the lip and gum a packet consisting of areca nut, calcium hydroxide (slaked lime), betel leaf, tobacco, and various flavorings.12 Several components of betel quid in addition to tobacco, such as areca nut, are known carcinogens (betel quid as well as areca nut are classified as Group I carcinogens by the International Agency for Research on Cancer [IARC]), and this mode of use can be especially damaging because the packets are often in contact with oral mucosa for long durations.13
Alcohol consumption. Heavier consumption of alcohol is associated with increased HNC risk. There is also a possibility of genetic susceptibility to HNC among certain heavy drinkers of alcohol that may predispose them to such cancers.7, 14 The high alcohol consumption that for some accompanies tobacco use might play a synergistic role in causing HNC as well.15
HPV infection. Incidence of HPV-related HNCs, specifically HPV-positive oropharyngeal cancer (OPC), has been growing quickly. HPV itself is widespread in the world’s population, making it a common risk factor. OPC is the most common HPV-positive cancer, with distinctly different demographics from non-HPV-associated OPC. Individuals with HPV-positive HNCs tend to be younger, healthier (fewer comorbid diseases than in long-term tobacco and alcohol users), and have different behavior- related risk factors, including more sexual partners and earlier ages of onset of sexual activity.
Currently there are more than 100 viral types of HPV. HPV 16 is considered to be responsible for the majority of HPV-associated cancers. There is evidence that vaccination against HPV 16 and 18 could prevent 90% of oral HPV infections within four years, but no data yet show if vaccination could translate into lower rates of OPC.3
Some sources conflate HPV-positive oral cancer with HPV-positive oropharyngeal cancer. At this time there is a significant difference in prognosis between HPV-positive and HPV-negative OPC. This is not the case for HPV-positive and HPV-negative oral cancers. The role of HPV in these cancers is not yet fully understood.16
Other viruses
The Epstein-Barr virus, a type of herpes virus ubiquitous in all human populations, is classified as a Group I carcinogen by the IARC due to its association with certain lymphoid and epithelial malignancies. It is also implicated as an etiologic agent in nasopharyngeal cancer.7, 17
The Hepatitis C virus may be associated with non-oropharyngeal cancers and with HPV-positive OPC. This is a recent finding which is still being confirmed.7, 18
Those with human immunodeficiency virus (HIV) are at increased risk of malignancy generally and for OPC specifically, with a two- to three-fold increased risk of head and neck SCC.7 This is likely associated with this population’s higher rates of concomitant HPV infection.19
HNC Staging Updates
HNC is staged using the tumor, node, metastasis (TNM) system. On January 1, 2018, the AJCC Cancer Staging Manual, Eighth Edition (AJCC 8) went into clinical effect1 and brought several important staging changes for HNC. The three most significant are:16 depth of invasion (DOI) in oral cancer was added to the T classification for oral cavity cancers; existence of extranodal extension (ENE) was added as a staging factor to the N category except for nasopharyngeal cancer and HPV-positive OPC; and a new and separate staging system was introduced for high- risk HPV-positive OPC.
DOI was added to the T category for HNCs as a better measure of tumor aggressiveness than overall tumor size, replacing tumor thickness. DOI is measured “from the horizontal line of the basement membrane of the adjacent healthy squamous mucosa until the deepest portion of tumor invasion.”20 Data over several decades have shown that DOI is a negative prognosticator in cancers of the tongue, buccal mucosa, and floor of the mouth.16 Replacing tumor thickness with DOI in the T classification is expected to yield better hazard discrimination.20
ENE was added to the N category as a factor in the clinical and pathological staging of HNCs in all anatomic sites, with the exception of HPV-positive OPC. Pathologic ENE is defined as extension of a metastatic carcinoma through the fibrous capsule of a lymph node. ENE is subdivided into microscopic (ENEmi), which extends 2 mm or less from the nodal capsule, and macroscopic (ENEma), which extends more than 2 mm or can be detected without a microscope. Clinical ENE is based on unambiguous clinical evidence, not only radiographic, of gross ENE.16
Staging criteria for OPC was the most significant category of changes. Staging criteria are now in two separate lists and are based on HPV status (positive or negative). HPV status is to be determined using immunohistochemistry (IHC), a technique for detection of protein markers that can aid in tumor classification and diagnosis of several cancers. In this case, IHC is used to detect p16, a tumor suppressor protein that overexpresses in the presence of transcriptionally active HPV. It has been found to be a good surrogate marker for the older polymerase chain reaction (PCR) test.21 Testing for p16 is more accessible, yields easy to interpret results, and costs significantly less to administer than PCR. The College of American Pathologists recommends that p16 IHC be reported as positive when there is “at least 70% nuclear and cytoplasmic expression with at least moderate to strong intensity.”36 Tumors that test negative for HPV are staged using Table 1, and those that test positive are staged using Table 2.1
Table 1. Oropharyngeal Cancer (p16 negative) |
|---|
T | Primary Tumor |
Tis | Carcinoma in situ |
T1 | Tumor ≤2 cm in greatest dimension |
T2 | Tumor >2 cm but not >4 cm in greatest dimension |
T3 | Tumor >4 cm in greatest dimension or extension to lingual surface to epiglottis |
T4 | Moderately advanced or very advanced disease |
T4a | Moderately advanced local disease: Tumor has invaded the larynx, extrinsic muscle of tongue, medial pterygoid, and hard palate or mandible |
T4b | Very advanced local disease: Tumor has invaded the lateral pterygoid muscle, pterygoid plates, and lateral nasopharynx or skull base or encases carotid artery |
N | Regional Lymph Nodes (Pathological, pN) |
N0 | No regional lymph node metastasis |
N1 | Metastasis in a single ipsilateral lymph node, ≤3 cm in greatest dimension and ENE(-) |
N2 | Metastasis in a single ipsilateral lymph node, ≤3 cm in greatest dimension and ENE(+); or >3 cm but not larger than 6 cm in greatest dimension and ENE(-); or metastases in multiple ipsilateral lymph nodes, none >6 cm in greatest dimension and ENE(-); or metastases in bilateral or contralateral lymph nodes, none >6 cm in greatest dimension and ENE(-) |
N2a | Metastasis in a single ipsilateral or contralateral node ≤3 cm in greatest dimension and ENE(+); or a single ipsilateral node >3 cm but not >6 cm in greatest dimension and ENE(-) |
N2b | Metastasis in multiple ipsilateral nodes, none >6 cm in greatest dimension and ENE(-) |
N2c | Metastasis in bilateral or contralateral lymph nodes, none >6 cm in greatest dimension and ENE(-) |
N3 | Metastasis in a lymph node >6 cm in greatest dimension and ENE(-); or metastasis in a single ipsilateral node >3 cm in greatest dimension and ENE(+); or multiple ipsilateral, contralateral or bilateral nodes, any with ENE(+) |
N3a | Metastasis in a lymph node >6 cm in greatest dimension and ENE(-) |
N3b | Metastasis in a single ipsilateral node >3 cm in greatest dimension and ENE(+); or multiple ipsilateral, contralateral or bilateral nodes, any with ENE(+) |
M | Distant Metastasis |
M0 | No distant metastasis |
M1 | Distant metastasis |
Source: AJCC 8
Table 2: HPV-Mediated Oropharyngeal Cancer (p16 positive) |
|---|
T | Primary Tumor |
T0 | No primary identified |
T1 | Tumor ≤2 cm in greatest dimension |
T2 | Tumor >2 cm but not larger than 4 cm in greatest dimension |
T3 | Tumor >4 cm in greatest dimension or extension to lingual surface of epiglottis |
T4 | Moderately advanced local disease: Tumor has invaded the larynx, extrinsic muscle of tongue, medial pterygoid, and hard palate or mandible or beyond |
N | Regional Lymph Nodes (Pathological, pN) |
pN0 | No regional lymph node metastasis |
pN1 | Metastasis in ≤4 lymph nodes |
pN2 | Metastasis in >4 lymph nodes |
M | Distant Metastasis |
M0 | No distant metastasis |
M1 | Distant metastasis |
Source: AJCC 8
Treatment
Current treatment recommendations for HNC call for a multidisciplinary approach, with the best outcomes for complex cases seen at high-volume treatment centers where expertise is concentrated.23
- Local disease (Stage I and II): 30% to 40% of patients will be diagnosed with local disease and can generally be cured with surgery and/or definitive (i.e., curative) radiation therapy (RT), which includes external beam RT and brachytherapy. Treatment often depends on accessibility of the anatomic site for surgery and associated patient morbidity.2 People with smoking and alcohol-related HNC, for example, are at risk for recurrence and second primary tumor occurrence, so alcohol and smoking cessation are important, as is close surveillance during follow-up. Surgical approaches may include: wide local excision for easily accessible sites; minimally invasive techniques such as transoral laser microsurgery (TOLM) for cancers of the larynx and hypopharynx; and transoral robotic surgery (TORS) for cancers of the oropharynx.23 The current standard of care requires intensity-modulated radiation therapy (IMRT) and image-guided radiation therapy (IGRT).23
Locally advanced disease (Stage III and IV): More than 60% of HNC patients are diagnosed with locally advanced disease, which carries a high risk of recurrence, distant metastases, and has poor prognosis. Treatment often includes a combination of surgery, radiotherapy, and chemotherapy.2 The oral cavity is usually accessible for surgery as a primary treatment. Because oral cavity tumors are generally aggressive, postoperative RT often combined with chemotherapy is common. For cancers of the pharynx and larynx consideration of preserving function is important, so TORS, TOLM, and chemoradiotherapy are often used. For cancers of the nasopharynx, RT is the primary treatment for locoregional disease as this area is not anatomically accessible for surgery. The addition of chemotherapy in more advanced-stage disease can improve the likelihood of survival. Salivary gland cancers are primarily treated with surgical resection for lower-grade tumors and additional RT for higher-grade carcinomas.23
HPV-positive OPC. This is currently being treated the same as HPV-negative OPC. Clinical trials are being conducted to look at deintensification of treatment for these patients.23
Recurrent and/or metastatic disease. This develops in about 65% of patients. Several clinical trials have shown a survival benefit of adding immune modulators to standard therapy.2
Survival Outcomes
The longest-running cohort study of HNC patients was a 2014 study which followed 1,657 individuals from British Columbia, diagnosed with SCC of the oral cavity, pharynx, or larynx, between 1986 and 1990, for up to 25 years. Overall survival (OS) was tracked and compared by cancer site (using AJCC 5 staging), treatment received, and risk factors. Overall survival at 5, 15, and 25 years for all sites in the aggregate was 64%, 46%, and 21%, respectively. Median age at diagnosis was 63 years, 74% of individuals were male, and 43% were diagnosed at stage III or IVA. Median overall survival length ranged from 1.4 to 8.7 years, with glottic larynx cancer having the longest and hypopharynx cancer the shortest.
Figure 1 shows percent OS at 2, 5, 15, and 25 years by anatomic site. Risk factors associated with poorer survival included: older age, male gender, later stage at diagnosis, and oropharyngeal or hypopharyngeal disease.24
Figure 1.
Overall Survival (%) of SCC HNC by site in Canadian Patients Diagnosed 1986-1990
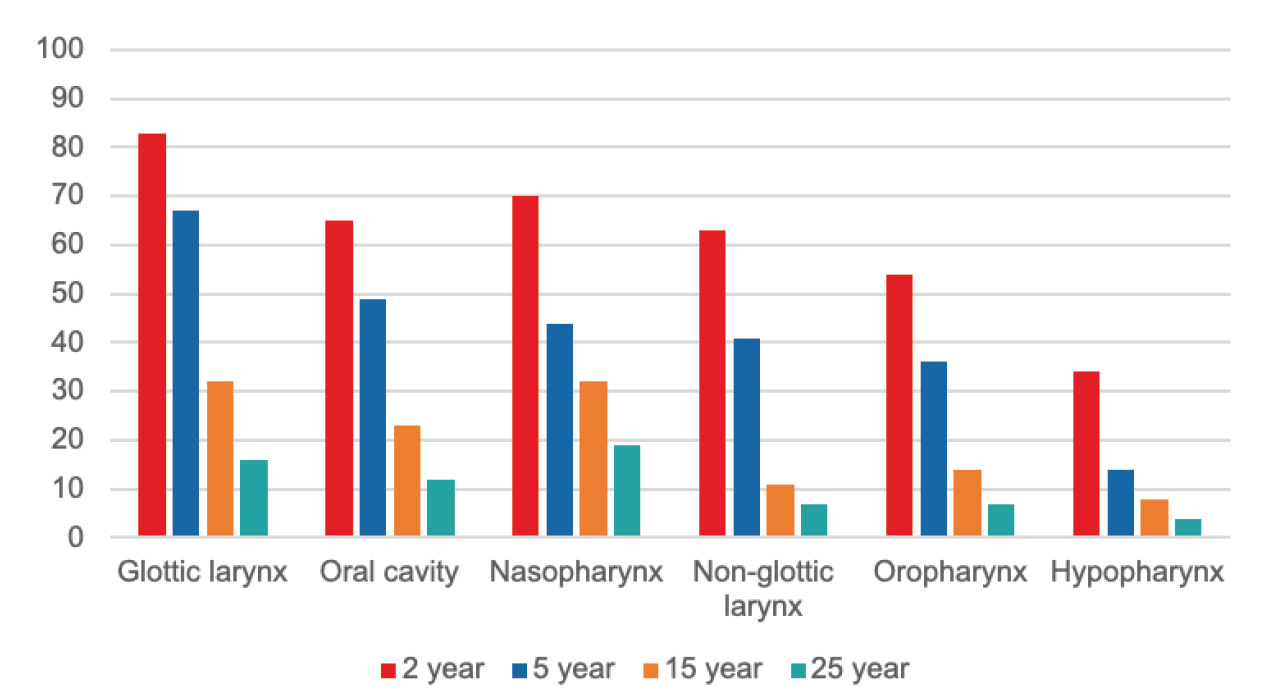
Source: Tiwana MS, et al. 25-Year Survival Outcomes for SCCs of the Head and Neck. Oral Oncology 201424
In a U.S. cohort from the Carolina Head and Neck Cancer Study,25 10-year survival rates of patients with SCC of the head and neck were examined in patients who had survived five years.
Patients with HNC (SCC) were compared to age-matched non-cancer controls, stratified by p16 status and smoking status. OS at 10 years was 87% for p16+ oropharynx, 56% for p16-oropharynx, 69% for oral cavity, 67% for larynx, and 51% for hypopharynx. Predictors of mortality included initial stage at diagnosis, anatomic site, smoking status, and p16 status, with p16+ individuals having more favorable prognoses.25
Survival for this cohort likely looks more favorable when compared to other studies because patients were selected who had already survived for five years and were followed for an additional five years, as opposed to following from time of diagnosis. For an estimate of five-year survival of U.S. individuals followed from time of diagnosis, see the SEER data in Table 3.26
Table 3: Five-Year Survival Rates for HNC by Site and Stage at Diagnosis (SEER 2009-2015) |
Cancer Site | Five-year Survival (Localized) | Five-year Survival (Regional) | Five-Year Survival (Distant) |
Salivary glands | 94% | 65% | 35% |
Nasopharynx | 82% | 73% | 48% |
Oral cavity and oropharynx | | | |
Lip | 92% | 60% | 28% |
Tongue | 81% | 68% | 39% |
Floor of mouth | 77% | 38% | 20% |
Nasal cavity and paranasal sinuses | 84% | 51% | 42% |
Larynx and hypopharynx | | | |
Supraglottis | 61% | 47% | 30% |
Glottis | 83% | 48% | 42% |
Subglottis | 60% | 33% | 45% |
Hypopharynx | 59% | 33% | 21% |
Source: Cancer.org26
Several other long-term cohort studies conducted in different global populations had similar results. In a 10-year study of prognostic factors for HNC survival in an Italian cohort (n=482), patients with primary SCC of the head and neck diagnosed in 2002-2012 were followed for up to 10 years post-diagnosis with median follow-up of 49 months. Five-year overall survival, combined across HNC sites, was 60.6%. By site, OS was 49.0% for oral cavity; 54.8% for oropharynx; 50.0% for hypopharynx; and 63.4% for larynx. Predictors of mortality included older age and more advanced tumor stage at diagnosis. Drinking eight to 14 alcoholic beverages per week was also a predictor of recurrence, and later stage at diagnosis and smoking for more than 40 years were predictors of a second primary HNC.27
In another study from Italy,28 five-year overall survival of HNC was calculated for 801 patients from five centers and predictors for overall survival evaluated. OS at five years was 64% for all HNC sites in aggregate, and survival by site was 55% for oral cavity, 53% for oropharynx, 41% for hypopharynx, and 71% for larynx. Predictors of poor survival were older age, higher tumor stage, high alcohol consumption, and for oral cavity cancer, combined therapy was associated with poorer prognosis. Higher tumor stage at diagnosis was also a predictor of recurrence and duration of smoking a predictor of second primary cancer.
Finally, a study examined a 1,829-patient cohort from the Scottish Audit of Head and Neck Cancer that were assessed 12 years after diagnosis. Overall survival was 26.3% across HNC sites and net survival was 41.4%. Predictors of mortality were anatomical site, age, cancer stage, treatment modality, World Health Organization (WHO) performance status, alcohol consumption, and smoking behavior.29
Additional Considerations
A separate discussion of several types of HNC is warranted, given differing predictors or prognoses. HPV-positive OPC has a better prognosis than HPV- negative OPC.30 HPV-positive OPC is now understood to be an entirely different entity than traditional smoking/ tobacco and alcohol-related OPC.31 In a study that looked at a U.S. cohort of 4,454 OPC patients where overall survival for HPV-positive and HPV-negative individuals was calculated separately using AJCC 7 staging, overall survival at four years for stages I to IV was 61.8%, 56.3%, 61.1%, and 55.8%, respectively, for HPV-negative OPC, and 90.1%, 86.1%, 87.0%, and 80.1%, respectively, for HPV-positive OPC. Significantly more favorable survival was demonstrated for HPV-positive OPC.32
The ICON-S study33 included 1,907 HPV-positive and 696 HPV-negative patients from seven centers in Europe and North America with a diagnosis of non-metastatic oropharyngeal cancer. It compared overall five-year survival for these patients using AJCC 7 staging and found that the staging performed poorly for HPV-positive patient stage separation. Using recursive partitioning analysis (a statistical method for multivariable analysis) and adjusted hazard ratio modeling, the study derived and tested new stage classifications for HPV-positive OPC. These new classifications were found to be a more valid way to stratify patients into stages and were incorporated into AJCC 8. Indeed, in the new classification system, 48% of HPV-positive stage III and IV cancers under AJCC 7 staging criteria would be stage I using AJCC 8 staging.16
Available data on survival outcomes for HNCs at salivary gland sites is limited due to that cancer’s rarity. Predictors of survival are also different for this cancer than for other HNCs. A 20-year retrospective cohort study reviewed pathology data on 75 U.S. patients diagnosed with salivary duct carcinoma from 1995 to 2014.34 Mean age at diagnosis was 66 years, 71% were male, and most primary tumors were in the parotid gland (83%), followed by the submandibular gland (12%). Histologically, 41% of the cancers were classified as carcinoma ex pleomorphic adenoma, 69% had perineural invasion, 58% showed extracapsular spread, 31% were ERBB2 gene positive, and 61% had vascular invasion. The median OS was 3.1 years. Features associated with poor survival included perineural invasion, vascular invasion, and extracapsular spread.34
Nasopharyngeal cancer (NPC) is rare in North America and Europe but more common in Asia. A cohort of 527 patients treated at a Chinese medical center between 2007 and 2011 with confirmed NPC without metastases was followed for up to five years. The median age of the cohort was 44 years, 75% were male, and 99% had non-keratinizing carcinoma. The findings included that five-year overall survival was 80.9%, and significant prognostic factors associated with poor survival included older age and higher stage.35
Conclusion
Head and neck cancers represent a diverse spectrum of tumors. Risk factors include tobacco use, excessive alcohol consumption, and infection with certain viruses such as HPV. In general, prognosis depends on age, gender, stage, and anatomic location of the tumor, among other factors. The risk of recurrence or a new primary tumor can be of concern under certain circumstances.
HPV-positive OPC is now understood to be an entirely different entity than the traditional tobacco and alcohol- related OPCs, with a prognosis better than for HPV- negative OPC. As it is staged so differently in AJCC 8 from before, it is important for underwriters and medical directors to understand that HPV status now drives the criteria by which this cancer is staged as well as the overall prognosis. A pathology report for OPC without an HPV status noted will likely be difficult to categorize, but this should be an unusual occurrence as HPV testing is now recommended for all patients with newly diagnosed oropharyngeal squamous cell carcinoma.36
The other important takeaway for underwriters is to appreciate how vast is the field of head and neck cancer, in terms of the complex anatomical areas involved. Different anatomical areas may be closely situated but can have very different cancers with different prognoses. Ensuring an HNC is correctly categorized and staged is of the utmost importance.