Musculoskeletal conditions are becoming a major burden on healthcare systems around the world.
They are affecting individuals as well as health and social care systems, with indirect costs a predominant factor. For arthritis alone, more than 100 types are currently known to exist.
Although arthritis has traditionally been considered a disease of older age, diagnosis of individuals between the ages 18 and 64 has been on the rise. It is estimated that more than one-third of all arthritis cases diagnosed from 2015 to 2017 were in this age cohort.1
In 2017, 92.1 million adults in the U.S. alone were either already diagnosed with arthritis or were reporting associated symptoms. And with the global population expected to rise by 25% in the next 30 years, further growth in arthritis cases appears imminent.2
Rheumatoid arthritis (RA), a progressive form of arthritis with associated inflammation, swelling, and pain in and around affected joints, is a chronic autoimmune disease. RA is commonly associated with progressive disability, systemic complications, early death, and high socioeconomic costs. It is unusual in that it can also affect other organs, such as the skin, blood vessels, heart, and lungs. Its estimated global prevalence is 0.46%, and approximately one in 12 women and one in 20 men are likely to develop RA in their lifetimes.2, 3
RA has a highly complex multi-phase pathophysiology which is influenced by risk factors such as the individual’s genetics, presence of infectious disease, and habits such as smoking and diet, resulting in progressive erosion and, in the most severe cases, systematic destruction of the affected joint(s).
As RA is an autoimmune disorder, lack of treatment or management may result in a more severe form of the disease, affecting other systemic organs. This aspect is known as “extra-articular involvement,” and is generally understood to indicate progression and poorer prognosis.
Diagnosing RA
Diagnosing RA and distinguishing it from other autoimmune disorders can be challenging. The diagnosis usually involves a holistic assessment that includes:4
Physical examination of affected joints
Blood tests such as:
Imaging studies (e.g., X-Rays, CT scans, MRIs)
Individuals with RA have at least one joint with a clinical diagnosis of synovitis. Synovitis is an inflammation of the synovial membrane, the connective tissue that lines joints such as the hip or knee, which cannot be explained as part of another disease.
The 2010 American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) 10-point classification system, detailed in Table 1 (below), requires a score of six or higher to diagnose RA.5
High blood titers of RF or ACPA are indicative of poorer functional and radiographic outcomes. However, it is important to understand that none of these tests is specific enough on their own to establish a definitive diagnosis of RA.
Table 1: Scoring and Diagnosing RA |
| Score |
Joint involvement | |
1 large joint | 0 |
2-10 large joints | 1 |
1-3 small joints (with or without involvement of large joints) | 2 |
4-10 small joints (with or without involvement of large joints) | 3 |
>10 joints (including at least one small joint) | 5 |
Serology (at least one test result needed) | |
Negative RF and negative ACPA | 0 |
Low-positive RF or low positive ACPA | 2 |
High-positive RF or high positive ACPA | 3 |
Acute-phase reactants (at least one test result needed) | |
Normal CRP and normal ESR | 0 |
Abnormal CRP or abnormal ESR | 1 |
Symptom duration | |
<6 weeks | 0 |
>6 weeks | 1 |
Source: Aletaha et al. 20105
Comorbidities Associated with RA
RA is associated with several comorbidities and is characterized by many complications. Table 2 (below) summarizes those commonly associated.2, 6-13
Table 2: Common Associated Complications |
System | Associated Complications |
Cardiovascular | Increased atherosclerosis risk Endothelial cell damage, which may predispose cholesterol deposition, leading to plaque formation and increased risk of stroke and myocardial infarction (MI). Ischemic heart disease risk is nearly 1.6x higher one to four years after initial RA diagnosis12 Endocarditis, pericarditis, left ventricular failure, valvulitis, fibrosis NSAIDs can increase risk of cardiac complications
|
Hematologic | |
Pulmonary | Interstitial lung disease (may progress to end-stage lung disease and in severe cases may require a lung transplant) Fibrosis (possible side effect of methotrexate and leflunomide) Pleural effusion Caplan’s syndrome (rheumatoid pneumoconiosis) – comparatively rare, seen among RA patients previously exposed to coal dust
|
Dermatologic | Rheumatoid nodules (occurs in 30% to 50% of RA cases) Vasculitis – in severe cases, ulcers may develop on legs and nail beds Thinning of skin due to corticosteroid use Skin sensitivity due to NSAIDs and methotrexate use
|
Ophthalmologic | Sjogren’s syndrome Episcleritis or scleritis (severe cases can progress to scleromalacia perforans) Uveitis (inflammation of the eye) – can lead to blindness if untreated Keratoconjunctivitis sicca (dry eye disease)
|
Musculoskeletal | |
Neurologic | Carpal tunnel syndrome Mononeuritis multiplex Myelopathy
|
Cancer | Lymphoma (rare) Non-melanoma skin cancer
|
Liver and kidney | Liver failure due to long-term acetaminophen use Kidney impairments due to use of cyclosporine, methotrexate, NSAIDs
|
Dental | |
A study which compared the presence of comorbid disorders among RA individuals to an age- and sex- matched population showed higher odds ratios (ORs) for disorders or diseases of these systems:
14
circulatory (12.7% vs. 9.4%)
endocrine (4.3% vs. 3.1%)
respiratory (6.5% vs. 3.8%)
In addition, higher prevalence of diabetes, cardiovascular, and respiratory diseases such as chronic obstructive pulmonary disease (COPD) was noted in individuals prior to their RA diagnoses. This indicates that individuals with RA may develop certain comorbidities before the onset of the disease, perhaps due to associated development of pre-diagnostic inflammatory processes.
Rheumatoid Arthritis and Mortality Trends
RA should be considered a systemic disease rather than one only affecting joints. Its impact on mortality is considerably different from some of the other arthritic conditions.
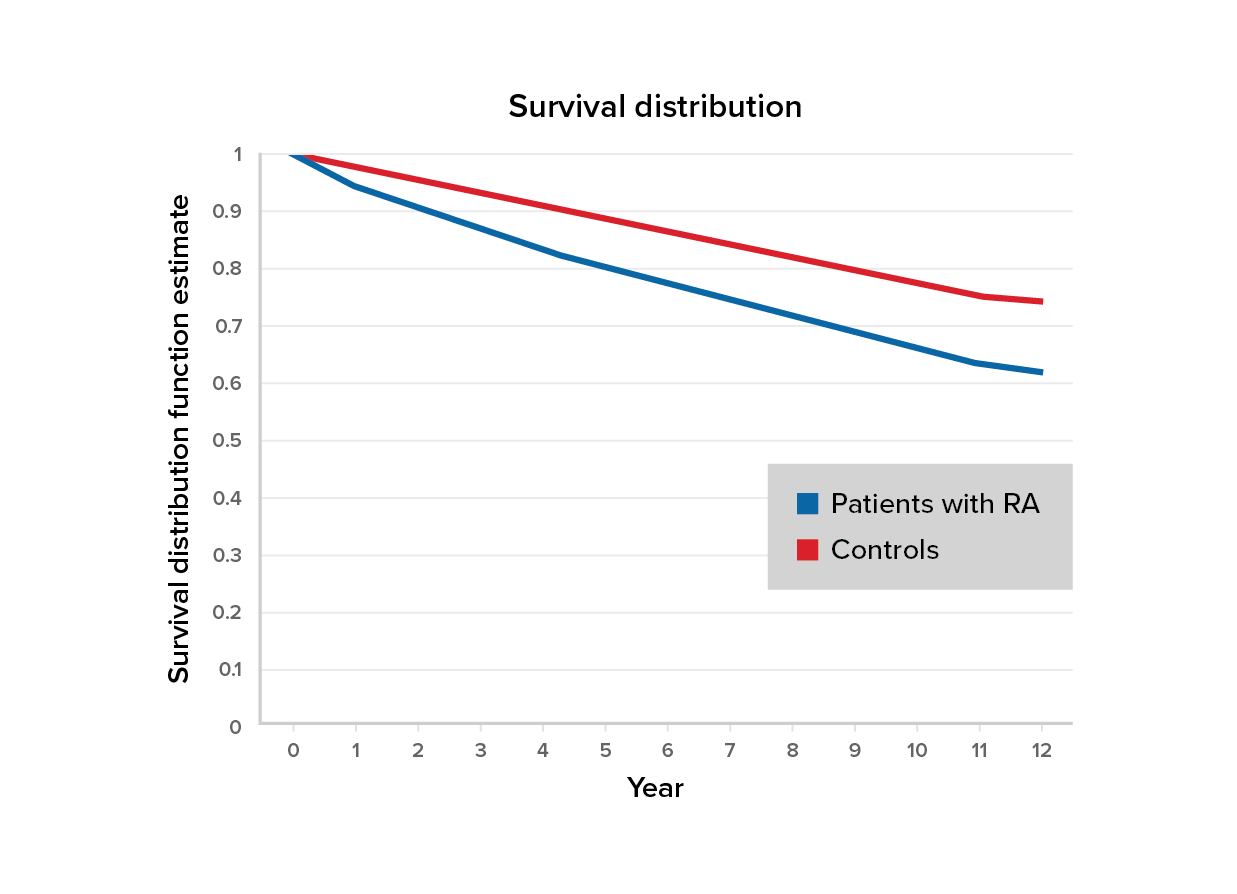
Figure 1:
Survival of RA patients vs. controls
Source: Loppenthin et al 14
According to Figure 1, a five-year survival rate of 80% (95% CI, 78-81%) was noted for individuals with RA versus 88% (95% CI, 88-89%) for control subjects (age- and sex-matched populations without RA).
Overall survival was persistently lower by less than 10% for RA patients compared with controls. After almost 12 years, the probability of survival among people living with RA was 61% (95% CI, 61-63%) compared to 74% (95% CI, 74-75%) for the controls.
An odds ratio (OR) of 1.41 (95% CI, 1.29-1.64) was observed for the cohort representing RA individuals compared with an age- and sex-matched population.14
The Impact of Rheumatoid Factor (RF)
Rheumatoid factors are proteins that are made by the immune system. Their presence or absence can help determine an RA patient’s prognosis.15 Patients with high levels of RF in their blood (more than 20 IU/ml) may be at higher risk of mortality and are prone to poorer prognostic outcomes. It must be understood, however, that high levels may be indicative of other autoimmune disorders such as Sjogren’s syndrome.
A study conducted among 603 individuals from the state of Minnesota (U.S.) revealed the difference in mortality between RF positive (RF+) and RF negative (RF-) individuals, benchmarked against expected mortality rates from 1970 to 2000. (See Figure 2, below.)15
Figure 2:
Mortality comparison – RF+ vs RF-
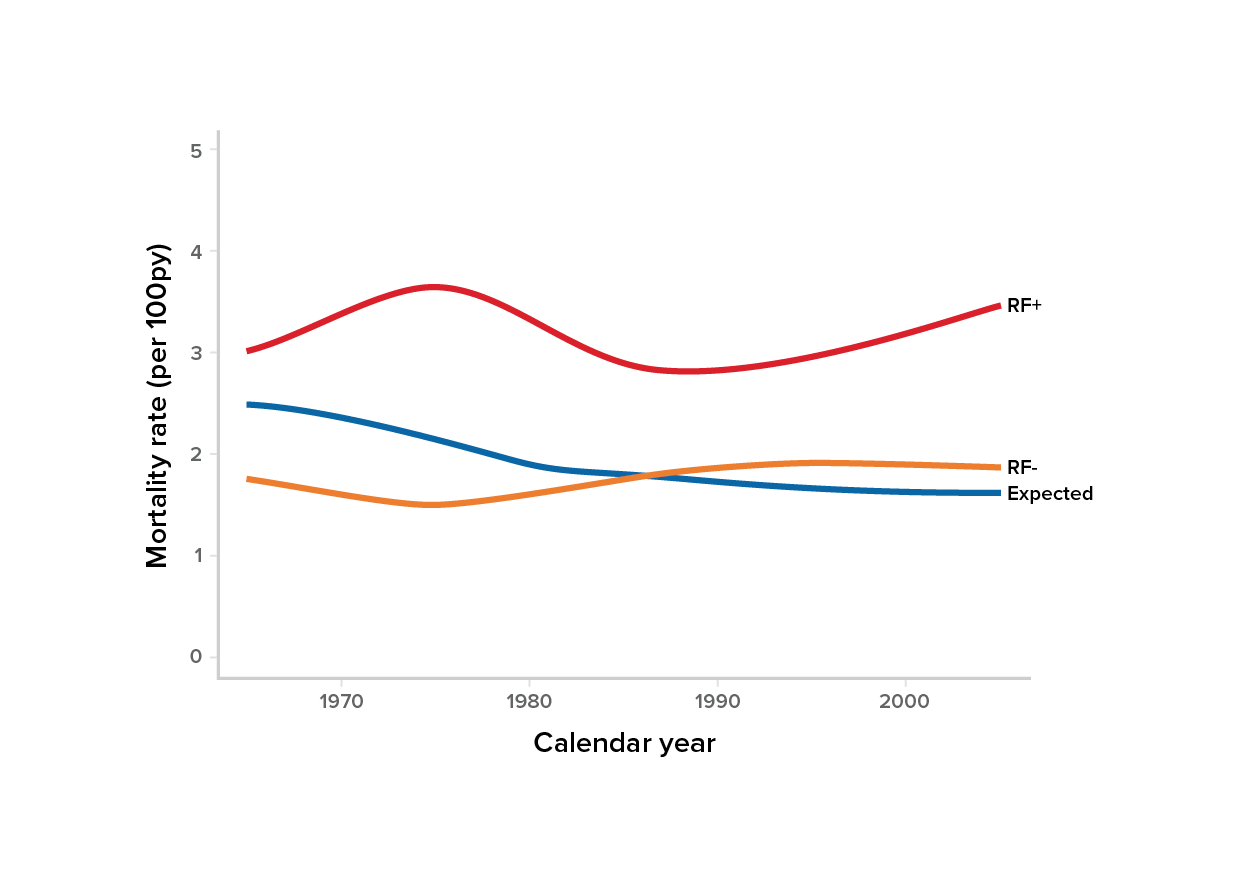
Source: Gonzalez, et al.15
According to the study, the mortality rate for the RF+ cohort was higher than expected compared to RFcohort. The standardized mortality ratio (SMR) for RF+ individuals was 1.81 (95% CI, 1.6-2.05), whereas the SMR for RF- individuals was similar to the expected mortality for both age and sex matched populations.
Other Factors
In addition to rheumatoid factor, other risk factors which may affect prognosis of RA are:16
Age: Those older than age 50 usually have poorer prognoses and higher mortality.
Education and socioeconomic levels: Those with high school diplomas or college degrees had lower mortality rates due to RA than those without, indicating that better awareness and access to treatment protocols may help in mortality reduction.
Other comorbidities: The presence of angina pectoris and hypertension doubled mortality risk for RA individuals compared to the general population, and impairments such as MI and stroke tripled the risk.
Smoking: Smoking not only increases the risk of RA but is also responsible for excess mortality among RA patients who smoke.
Treatment Modalities
Treatment of RA is often multidimensional. Disease- modifying medicines along with steroids and analgesics are recommended in most cases. Table 3 (below) summarizes the treatments advised for RA.17, 18
Disease-modifying anti-rheumatic drugs (DMARDs) are generally the drug(s) of choice for treating RA. They block the effects of chemicals released when the immune system attacks the joints and causes damage to nearby bones, tendons, ligaments, and cartilage.
csDMARDs (Conventional DMARDs): These are primarily administered during Phase 1 of an RA treatment regimen. Methotrexate is usually the drug prescribed, often in combination with another csDMARD and a short course of corticosteroids to reduce inflammation and relieve pain. If methotrexate is contraindicated, leflunomide or sulfasalazine may be prescribed.
bDMARDs (Biological DMARDs): These are TNF inhibitors, T-cell costimulatory inhibitors, and IL-6 receptor inhibitors. They are usually administered in Phase 2 of a treatment regimen if Phase 1 treatments have not been adequate or effective.
tsDMARDs (Targeted synthetic DMARDs or JAK inhibitors): These newly developed drugs are used in combination in bDMARDs during Phase 2 treatment as well as in different combinations for Phase 3 treatment. These drugs are indicated when bDMARDs fail to achieve desired Phase 2 outcomes.
Analgesics such as NSAIDs are often useful in relieving RA pain while also reducing joint inflammation, although they will not prevent RA from worsening. Long-term NSAID usage may also lead to cardiac complications.
Corticosteroids are usually prescribed for short-term pain relief. They are commonly used as adjuvants – for example, while waiting for DMARD medicines to take effect or during an RA flare-up in Phase 1 treatment. Long-term corticosteroid usage can lead to multiple complications such as osteoporosis and muscle weakening and is advocated only for extreme cases.
Surgery is for cases where joint degeneration is substantial and is limiting a patient’s activity in ways that have considerable impact on activities of daily living (ADLs).
Physiotherapy and occupational therapy are frequently recommended to maintain joint range of motion, strength, conditioning, and dexterity.
Nutritional supplements such as calcium and vitamin D may help prevent osteoporosis, and fish oil may reduce joint pain and stiffness.
Table 3: RA Treatments |
Treatment Class | Examples |
Disease Modifying Anti-Rheumatic Drugs (DMARDs) | Conventional synthetic DMARDs (csDMARDs)
• methotrexate, hydroxychloroquine, sulfasalazine, leflunomide, gold salts
Biological DMARDs (bDMARDs)
• TNF inhibitors – etanercept, adalimumab, golimumab, infliximab
• T cell costimulatory inhibitor – abatacept
• IL-6 receptor inhibitors – tocilizumab, sarilumab
Targeted synthetic DMARDs (tsDMARDs)
• JAK inhibitors – tofacitinib, baricitinib, upadacitinib |
Analgesics | Non-opioid analgesics
Topical agents
Opioid analgesics |
Anti-inflammatories | Non-steroidal anti-inflammatory drugs (NSAIDS) Glucocorticoids – systemic and intra-articular |
Surgical procedures | Arthroscopy Joint replacement surgery |
Non-pharmacologic and non-surgical | Patient education
Psychosocial interventions
Physiotherapy
Occupational therapy
Orthotic devices Nutritional and dietary counseling Podiatry |
Sources: Smolen, et al.18, Medscsape.com23, Uptodate.com24
Remission
The goal of RA treatment is to achieve remission. Various definitions of remission are in use, but the ones listed below, which are based on those used by ACR and EULAR, are the most widely accepted.
Comprehensive Disease Control (CDC): This is defined as achievement of stringent control of the signs and symptoms of inflammation, according to Disease Activity Score 28 (DAS-28). DAS-28 is an index that measures disease activity in patients based on an examination of their joints, their ESR or CRP results, and an overall assessment of their health.
CDC is considered to have been achieved when the following criteria are met:19
A DAS-28 score of <2.6
Normal physical function, assessed using the Health Assessment Questionnaire Disability Index (HAQ-DI), and obtaining a score of <0.5.
Absence of progression as measured radiographically, and a modified total Sharp erosion score (mTSS) of <0.5.
Sustained remission: The phrase “sustained remission” is another commonly used term to describe remission in RA patients. The more commonly used definition of remission is the ACR/EULAR definition, which depends on DAS28 and uses ESR or CRP results as well as more stringent DAS-based criteria such as the Simplified Disease Activity Index (SDAI) and the Clinical Disease Activity Index (CDAI) to measure RA disease activity. A minimum of six months is a designated targeted sustained remission period. However, the definition of sustained remission remains flexible.20 Indeed, a 10-year follow-up study of RA patients observed that only 14% had achieved sustained drug-free remission.
A Brief Look: Future RA Treatments, Management
Guidelines for RA treatment and management are reviewed periodically by ACR and EULAR. As newer methods, including some alternative therapies, continue to emerge, some traditional methods are being either replaced or restructured.
Recent study findings indicate that:
Minimizing use of corticosteroids such as prednisone may be beneficial due to deleterious side effects from long-term use.
Switching between DMARDs is neither advisable nor preferred. If an RA patient starts on a csDMARD, for example, continuing on it long-term showed better prognostic outcomes.
Using a bDMARD or tsDMARD may be more effective than multiple doses or a triple therapy csDMARD regimen (methotrexate, hydroxychloroquine, sulfasalazine).
Continuing current medications is also recommended over tapering. Tapering can only be considered for people who have met their treatment goals consistently for at least six months.
Other advances:
Inflamation is associated with degenerative neurological conditions such as Alzheimer’s disease, and research has determined that RA can increase the risk of certain dementias. However, more recent studies have shown that those who took bDMARDs or tsDMARDs had a 17% reduced risk of dementia compared to those taking csDMARDs.
A new biomarker is improving the ability to predict cardiovascular disease risk in RA patients. UCLA researchers have found that RA patients with the lowest levels of low-density lipoprotein (LDL) had a 2.8 times higher risk of obstructive coronary plaques.21 This is referred to as the “lipid paradox,” as it is contrary to the general understanding of the impact of LDLs on coronary plaque.
A multi-biomarker disease activity (MBDA) score, also known as a Vectra score, is used to check levels of inflammation beyond CRP or ESR testing and help predict heart disease risk in RA patients.21
A new therapy for complex RA cases focuses on stimulation of the vagus nerve to reduce inflammation. Currently it is being studied in individuals who are not responding well to inflammation-lowering medications. This treatment will require larger study cohorts to better understand its long-term benefits.
New remission approaches:
Definitions of RA remission are constantly being updated. One recent update suggested that if RA patients are using combination therapy (e.g., a csDMARD plus a bDMARD), and if sustained remission was achieved, tapering should be considered for the csDMARD but not the bDMARD.
A Canadian study determined that early RA diagnoses may lead to better prognoses, milder disease, and possible long-term remission. Factors associated with lower remission rates included: female gender; having more comorbidities; being RF+; and smoking. The research underlined the importance of careful monitoring of individuals with RA who are at higher risk for transient (or partial) remission. Over a 24-month follow-up period, the study found that:22
55% of patients in the study achieved remission
47% of patients who achieved remission sustained it for 12 months or more
40% sustained remission for up to 24 months
Conclusion
RA is a complex disorder with a myriad of associated risk factors, complications, comorbidities, and extra-articular manifestations. Traditional drugs such as analgesics and DMARDs continue to be treatment mainstays. Research into new therapies is ongoing, but long-term remission remains elusive.
It is essential that underwriters, claims adjudicators, and insurance medical directors understand all aspects of RA’s progression as well as what constitutes adequate responses to its treatments, as the holistic risk profile contributes to subsequent morbidity and mortality risk.