Amid a rapidly evolving landscape with many trials underway, underwriters must carefully monitor this class of medications and prepare for additional changes to come.
News about obesity-combating drugs that target receptors such as GLP-1 is coming at us at warp speed. In just the past few months, since submitting this article to On The Risk, preliminary study results have come through showing that these drugs have efficacy in reducing risk of major fatal and nonfatal cardiovascular events for people with Type 2 diabetes. And that’s not all: Trials are also under way to examine possible efficacy for Alzheimer’s disease, Parkinson’s disease, certain cancer-related chronic pain, and neurological conditions such as schizophrenia. Additional studies seek to determine if these drugs could work for obesity due to genetic mutations, a particular factor in childhood cases.
News about new receptor agonist (RA) drugs in the pipeline is also emerging rapidly. One of the newest such drugs, retatrutide, which is still considered experimental, is showing substantial promise to improve weight loss rates from the current average of 15%-17% of starting weight to as high as 25% after one year. Trials are also under way for additional GLP-1 RA drugs as well as two targeting genes shown in the past to affect weight. And a just-released study from Found.com determined that 26% of the respondents – all large employers (>5,000 employees) – plan, in the next 12 months, to incorporate coverage for anti-obesity medication (or other form of weight-loss monitoring) in their benefits.
Although these drugs are highly promising in the ongoing quest to find reliable treatments for the obesity pandemic, the attributable positive side effects could simply be a function of weight loss’s beneficial metabolic impact. Negative side effects to these drugs exist as well, so careful monitoring is important. Still, progress in tackling obesity promises to yield significant health and mortality benefits. And although most health insurers worldwide have specific exclusions for obesity treatments, insurers would do well to keep track of the latest developments, both around these drugs and in how best to approach coverage and pricing.
Wegovy, Trulicity, Ozempic… Oh My!
Recently, it has become difficult to avoid the buzz – print ads, commercials, social media postings, and general discussion – about the new weight loss drug Wegovy®, a new indication of semaglutide also approved for treatment of Type 2 diabetes under the brand names Ozempic® and Rybelsus®. Whether it is discussion about drug shortages,1 side-effects, or videos showing weight loss results, this class of drugs has been big news in the last year. One analysis from February 2023 estimates there was an increase of over 2000% in prescribing of semaglutide (Ozempic, Rybelsus, Wegovy) and tirzepatide (Mounjaro®) for weight management between 2019-2022 (tirzepatide was approved in 2022). There was a 259% increase in prescriptions between 2021 and 2022 alone. In this analysis, one-fourth of prescriptions were for individuals without diabetes.2 Finding a new, effective treatment for obesity has significant public health implications given the prevalence of overweight and obesity and associated comorbidities in the United States.
Obesity in the United States
In the United States, the prevalence of obesity has been increasing over the last several decades. Approximately 30% of adults are classified as overweight (BMI 25-29.9), 42% as obese (BMI 30-40) and 9% as severely obese (BMI ≥40). Children in the U.S. also have a high prevalence of obesity, with a combined 41% of children being classified as either overweight or obese. Comorbidities associated with obesity affect both morbidity and mortality and include increased risk for Type 2 diabetes, hypertension, cardiovascular disease, stroke,3 musculoskeletal conditions, liver disease, gallbladder disease, some cancers, and sleep apnea. The aggregate medical costs of obesity in the United States was estimated to be over $260 billion in 2016.4
Figure 1. Trends in Age-Adjusted Obesity and Severe Obesity Prevalence Among Adults Ages 20 and Over: U.S., 1999-2000 Through 2017-2018.
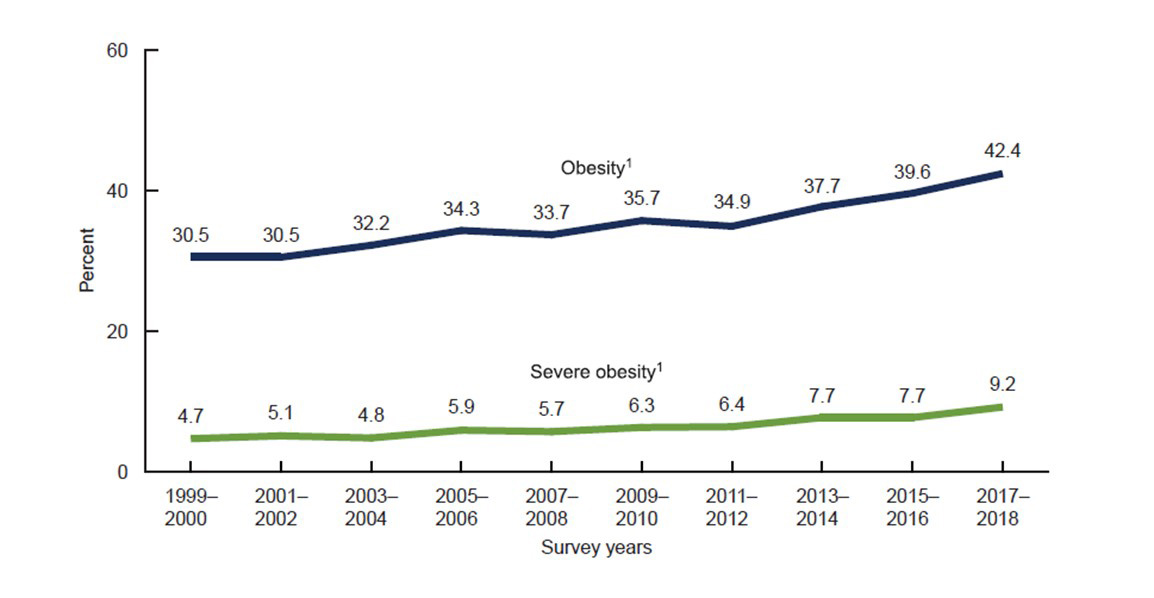
Note: Estimates were age adjusted by the direct method to the 2000 US Census population using the age
groups 20-39, 40-59, and 60 and over.
Source: www.niddk.nih.gov/health-information/health-statistics/overweight-obesity
Figure 2. Trends in Obesity Among Children and Adolescents Ages 2-19 Years, By Age: U.S., 1963-1965 Through 2017-2018.
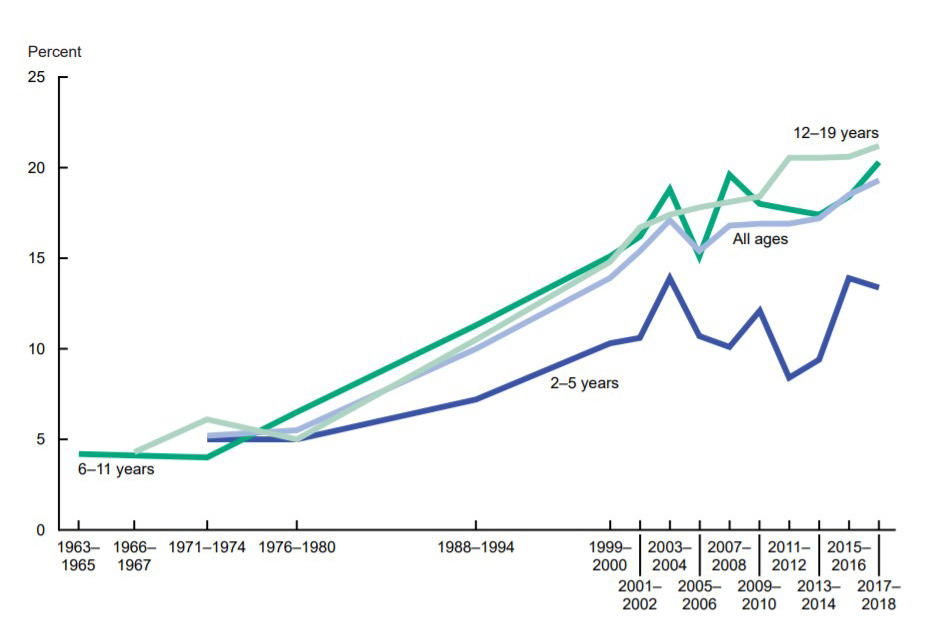
Note: Obesity is defined as body mass index (BMI) at or above the 95th percentile from the sex-specific
BMI-for-age 2000 CDC Growth Charts.
Source: www.niddk.nih.gov/health-information/health-statistics/overweight-obesity.
Glucogen-like peptide-1 receptor agonists (GLP-1 RAs)
The class of drugs getting so much attention for weight loss are GLP-1 receptor agonists (GLP-1 RAs). They are synthetic analogues of GLP-1 that act to control diabetes in four main ways: they stimulate insulin release in response to glucose, reduce hepatic glucose production by inhibiting glucagon release, increase satiety, and slow gastric emptying. Unlike natural GLP-1, which is degraded very quickly, the synthetic version tends to stick around longer.5
GLP-1 is an incretin hormone produced in the L cells of the small intestine and released upon eating. GLP-1 stimulates glucose-dependent-insulin release from the pancreas islets. GLP-1 receptors are located throughout the GI system in the pancreas, pancreatic ducts, and gastric mucosa and outside the GI system in the kidney, heart, lungs, skin, hypothalamus, and immune cells.6
Since the first GLP-1 RA medication became available in 2005, several additional medications have been added to this class. They were initially developed as treatments for Type 2 diabetes7 and have demonstrated significant reductions in A1c in individuals with Type 2, minimal risk of hypoglycemia, and favorable effect on weight. Additionally, dulaglutide, liraglutide, and semaglutide (injectable) have shown evidence for reduction in major cardiovascular events. Use of the GLP-1 RAs may be limited by high cost, need for injection of most of the agents, and adverse GI effects in some individuals.8
Review of available GLP-1 RAs in the United States
Table 1. Summary of GLP-1 RAs available in the United States
| Generic Name | Brand Name(s) | Formulation | FDA Approval | Highlights | Contraindications |
| semaglutide9 | Wegovy10 | 2.4 mg weekly injection | Approved 6/2021 for weight management in adults and children age 12+. | Approved for weight loss. Approved in teens. | Personal or family history of medullary thyroid cancer or MEN 2A. |
| Ozempic11 | 0.5, 1.0, 2.0 mg weekly injection | Approved 12/2017 to improve blood glucose in adults with Type 2 diabetes and to reduce major CVD events in people with Type 2 and CVD. | Approved for both Type 2 and to reduce major CVD events in Type 2. |
| Rybelsus12 | 7.0, 14.0 mg daily tablet | Approved 9/2019 for blood glucose control in adults with Type 2 diabetes. | First oral GLP-1 drug available in the U.S. |
| tirzepatide13 | Mounjaro | 2.5, 5, 7.5, 10, 12.5, 15 mg weekly injection | Approved 5/2022 to improve blood glucose control in adults with Type 2 diabetes. | Approved 5/2022 to improve blood glucose control in adults with Type 2 diabetes. | Personal or family history of medullary thyroid cancer or MEN 2A. |
| liraglutide | Victoza®14-16 | 1.2, 1.8 mg daily injection | Approved 1/2010 (original approval) for Type 2 in adults; Additional approval 8/2017 to reduce CVD events in type 2; Approved 6/2019 to treat patients 10+ with Type 2 diabetes. | Approved to treat Type 2 in kids ages 10 and up. Also approved to reduce CVD events in adults with Type 2 diabetes. | Personal or family history of medullary thyroid cancer or MEN 2A. |
| Saxenda®17,18 | 3 mg daily injection
| Approved 12/2014 for chronic weight management in adults; Approved 12/2020 for kids age ≥ 12. | Approved for weight loss in kids ages 12-17. |
| dulaglutide | Trulicity®19-21 | .75, 1.5, 3.0, 4.5 mg weekly injection | Approved 9/2014 to improve glycemic control in adults with type 2 diabetes and to reduce risk of CVD events in adults with Type 2; The 11/2022 approval was extended to kids age 10+ for Type 2 diabetes. | Approved for both Type 2 diabetes and to reduce major CVD events in Type 2 diabetes. Approved for use in kids ages 10 and up for Type 2 diabetes. | Personal or family history of medullary thyroid cancer or MEN 2A. |
| exenatide | Byetta® 22,23 | | Approved 4/2005 as adjunctive therapy for blood sugar control in patients with Type 2 diabetes who have not achieved control with metformin and/or a sulfonylurea; Approved 11/2009 as standalone treatment for Type 2 diabetes; Approved 10/2011 for use with insulin glargine. | This was the first approval in the class. Can be used with insulin. | Personal or family history of medullary thyroid cancer or MEN 2A. |
| Bydureon BCise® 24,25 | 2 mg extended release weekly injection | Approved 1/2012 for adults with Type 2 diabetes (injectable suspension); Pre-filled, single-use pen injector approved 3/2014; Approved 4/2018 as add-on therapy for use with basal insulin in adults with Type 2 diabetes and inadequate glycemic control; Approved 7/21 for ages 10+. | First weekly treatment for Type 2 diabetes. Can be used with insulin. Approved for Type 2 diabetes for kids ages 10+ as of 7/2021. |
| lixisenatide 26,27 | Adlyxin® | na | Approved 7/2016 for adults with Type 2 diabetes; Discontinued 1/2023.
| Discontinued 1/1/23 in U.S. market for business reasons. Soliqua – a formulation that contains insulin glargine and lixisenatide – is still available.28
| na |
Use for Weight Loss
While the GLP-1 RAs were initially developed to treat Type 2 diabetes, their weight-loss effects are currently getting more attention. All have shown some effect on weight, though their effectiveness varies (see table 2). So far, only semaglutide (Wegovy) and liraglutide (Saxenda) have FDA approval as therapy for weight loss. Both Wegovy and Saxenda are approved for teens (ages 12 and older) and adults, however, Wegovy is a weekly injection while Saxenda is a daily injection. In terms of efficacy, The STEP 8 randomized clinical trial compared use of semaglutide 2.4 mg weekly injection with liraglutide 3.0 mg daily injection in overweight and obese adults. After 68 weeks, those in the semaglutide group achieved a mean weight change of -15.8% and those in the liraglutide group a mean weight change of -6.4%. Both led to weight loss, but semaglutide led to greater weight loss.29 Tirzepatide is not currently FDA approved for weight loss but has received the FDA Fast Track designation30 to accelerate the path to approval. In a phase 3 trial31 of obese adults receiving tirzepatide weekly for 72 weeks at doses of 5-15 mg, the mean weight change was -15% across groups, with 85% of participants reducing their weight at least 5%. It is important to note that because all the medications in this class have effects on weight, there is evidence that they are being prescribed off label for weight loss.32
Table 2. Comparison of GLP-1 RAs: Effect on Weight Based on Phase III Studies*
|
| Drug | Effect on Weight Compared to Other GLP-1 RAs |
| dulaglutide (Trulicity) | Intermediate |
exenatide
Twice daily (Byetta)
XR (Bydureon BCise) |
Low
Low |
| liraglutide (Saxenda, Victoza) | High |
| lixisenatide (Adlyxin) # | Low |
semaglutide
Oral (Rybelsus)
Injection (Ozempic, Wegovy) |
Highest
Highest |
*Adapted fromTrujillo et al. 2021.32 #Discontinued in U.S. Market in 2023.26 Note – analyses for this table did not include tirzepatide.
What does all this mean for underwriting?
Overweight and obesity are very common in the U.S., and their comorbidities are associated with some of the most common causes of increased morbidity and mortality.33 While long-term data is lacking for these treatments, reduction in overweight and obesity should theoretically lead to improvement in downstream outcomes. However, this is speculative, and more research is needed over the next decade to study the effectiveness of these therapies over the long term. Currently, in underwriting, it is important to understand that use of this class of medications does not necessarily indicate that a PI is a Type 2 diabetic. It is also important to note that there is a warning about using these medications in individuals at risk for medullary thyroid cancer or MEN 2A. This is a rapidly evolving landscape with many trials underway as drug companies seek indication for weight loss (see tirzepatide), so underwriters will continue to see more of this class of medications with additional changes to come.





