End-stage kidney disease (ESKD), the most serious and dramatic manifestation of kidney disease, is today a worldwide public health issue, especially in developing countries. In developed countries, advances in the main treatment modalities – dialysis and transplant – have significantly improved life expectancies.
Indeed, innovative transplant surgical techniques, improved immunological risk assessment of donors and recipients, and better and more sophisticated immunosuppressive drugs have made renal transplantation a more effective treatment for patients with ESKD than long-term dialysis.
For underwriters, correctly assessing the potential mortality and morbidity risk of applicants who are past renal transplant recipients involves understanding a broad range of issues related to kidney disease, current transplantation science, long term graft success, post-transplant survival predictors, and the common causes of death in kidney transplant recipients.
Introduction
Since the middle of the last decade, incidence rates for ESKD have stabilized for the world’s more affluent countries, but have continued to increase for developing countries. Higher-income countries’ renal health priorities, which are aimed at improving the detection of chronic kidney disease (CKD) in the hopes of delivering treatments to slow its progression, likely play a role in limiting the progression to Stage 5 CKD (ESKD).2 In developing countries, however, diabetes, hypertension, and communicable diseases that can lead to acute kidney injury all play important roles in the rising rates.2
Part of the reason is continued treatment inequities, which need to be addressed. In addition, especially in tropical nations, environmental factors such as the higher rates of bacterial, viral, and parasitic infections found in these countries contribute to a glomerular disease prevalence 2.5 times higher than that of developed countries.
CKD and ESKD – Basics
Chronic kidney disease (CKD) is diagnosed using three prognosticators: renal function (as indicated by estimated glomerular filtration rate [eGFR]), imaging results (whether ultrasound, CT, or MRI) showing kidney damage, and the proteinuria/creatinine (or albumin/creatinine) ratio.
The National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative (KDOQI), which provides evidence-based guidelines for all stages of CKD, currently recommends using serum creatinine concentration, expressed by the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) creatinine equation, to calculate eGFR. Individuals with an eGFR of <60 ml/min/1.73m2 for three months, according to this guideline, are classified as having CKD, irrespective of the presence or absence of kidney damage. ESKD, which is Stage 5 CKD, is defined as an eGFR of <15 ml/min/1.73m2. Those at this eGFR level are candidates for permanent renal replacement therapy.
ESKD is considered to be caused primarily by diabetes, hypertension, glomerulonephritis (GN), and cystic diseases. Communicable diseases can also be causes. In the U.S., the most common ESKD etiologies are diabetes, hypertension, and GN. ESKD patients with GN as the primary cause are considered to have better prognoses than those whose ESKD is from other causes. However, these are the “primary” diagnoses for ESKD, assigned by the treating physician at the start of treatment. Quite commonly, the original etiology of the disease may be difficult to discern as renal patients are, unfortunately, frequently asymptomatic until they reach more advanced CKD stages.4
The convention of assigning only one primary cause to any ESKD case may be limited. This theory is supported by evidence that in many cases of ESKD attributed to hypertension, for example, preceding clinical features highly suggestive of renal parenchymal disease (moderate to severe proteinuria and hematuria) might have also been evident. In addition, ESKD ascribed to hypertension – usually a clinical diagnosis – could be wrong. The patient’s disease could have actually been an unrecognized primary glomerular disease, ischemic nephropathy or analgesic nephropathy.
The practice of grouping all patients with ESKD due to GN as a single clinical entity has not been seriously challenged, although clinicians have long recognized the heterogeneous nature of GN in the non-ESKD setting.9 A recent U.S. study illustrates the imprecision and heterogeneity of most of the categories used to describe causes of ESKD.14 Using Medicare administrative claims data from the United States Renal Data System, which collects, analyzes and distributes information about CKD and ESKD in the U.S., patients with GN-caused ESKD were classified into the following six GN subtypes:
- focal segmental glomerulosclerosis (FSGS)
- IgA nephropathy (IgAN)
- membranous nephropathy
- membranoproliferative glomerulonephritis (MPGN)
- lupus nephritis (LN)
- vasculitis
Large differences in patient mortality, as measured by the adjusted mortality hazard ratio (aHR), were found among the six groups. After adjustment for demographics and comorbidity, patients with IgAN (also known as Berger’s disease) had the fewest comorbidities and the lowest mortality. The next lowest were patients with membranous nephropathy (aHR 1.23), FSGS (aHR 1.37) and MPGN (aHR 1.38). The highest aHRs were in patients with LN (aHR 1.75) and diabetes (aHR 1.73), followed by vasculitis (aHR 1.51). Patients with IgAN were also found to have better survival rates than patients with autosomal dominant polycystic kidney disease (ADPKD), who are often considered to have among the best prognoses among patients with ESKD (aHR 1.22).6
Although this research partly illustrates the tendency for GN to become inactive and undifferentiated as kidney failure progresses to end stage (as may have occurred in our case study), it also raises questions, as there is no assurance that the diagnosis was based on biopsy. Also, an important prognostic factor is disease activity at onset of ESKD. This could be inferred by the presence or absence of immunosuppressive treatments given upon an ESKD diagnosis.
Transplantation
Transplantation has been an accepted ESKD treatment for the last 60 years. CKD patients are generally educated soon after their diagnosis about the natural progression of their disease, the different types of dialysis available, and the renal transplantation option.
The best evidence from observational data show that transplantation confers a mortality benefit in addition to improved quality of life compared with maintenance dialysis.
Indications for transplantation for patients with ESKD include:
- severe metabolic acidosis
- hyperkalemia (elevated potassium levels)
- clinical manifestations such as pericarditis, encephalopathy, and intractablevolume overload
- declining nutritional status
- a glomerular filtration rate of 5-9 ml/min/1.73m² in otherwise asymptomatic adults
Kidney donors can either be related or not to the recipient, and can be living or deceased as well. The degree of a transplant recipient’s immune response depends partly on the degree of genetic difference between an organ and its recipient. The human leukocyte antigen (HLA) system genes (or gene complex), which governs compatibility, are located on the short arm of the chromosome 6, and encode the major histocompatibility complex (MHC) proteins. These proteins are divided into class I
(HLA A, B and C) and class II (HLA DR, DQ and DP) antigens and are responsible for regulating the human immune system. The role of these proteins is to present peptides to T-cells, enabling them to recognize and eliminate “foreign” cells. The most common form of acute rejection of grafts involve a recipient’s T-cells (i.e. adaptive immune response) becoming activated against the donor’s MHC antigens.
Long-term renal graft survival has been shown to be closely related to the number of HLA mismatches at the time of transplantation.20 In our case study, the applicant had a low Panel Reactive Antibody (PRA) score and was fortunate to receive a kidney from a closely matched living donor.
Immunosuppressants
The first successful immunosuppressant was the purine analogue azathioprine. Since then, many other agents have been approved. They are currently classified into two categories: antirejection induction agents (powerful antirejection medicine used at the time of transplant, e.g. basilixumab, rabbit antithymocyte globulin, and alemtuzuma); and maintenance immunotherapy agents (e.g. prednisone, azathioprine, mycophenolate mofetil, cyclosporine, tacrolimus, sirolimus, and belatacept).
Choosing the correct immunosuppressive therapy involves balancing the risk of graft injury against the risks of drug toxicity, infection, and malignancy.7 Post-transplant immunosuppression therapy is normally a combination of agents, with the protocols developed following similar principles to antimicrobial and anti-neoplastic chemotherapy.
The intensity of the immunosuppression need is not constant. High levels are generally required right after the transplant, and can later be reduced to a maintenance level. Immunosuppression must, however, continue for the life of the recipient, for if it is stopped, rejection and then graft loss generally occurs. There is no optimal maintenance immunosuppressive protocol as yet, but the most common regimen used today in kidney transplantation is a CD25 monoclonal antibody such as basiliximab, followed by a combination of tacrolimus, mycophenolate mofetil, and steroids.7
Side effects of the immunosuppressors can also be an issue, as well as the higher risk of recipient infection and malignancy due to immunosuppression. Infection is the most common cause of their first-year mortality and morbidity. Recipients are at particularly high risk for non-Hodgkin lymphoma, Hodgkin lymphoma, Kaposi’s sarcoma, and infectious cancers of the liver, stomach, oropharynx, anus, and vulva.5
Assessment and Management of Renal Transplant Recipients
A kidney recipient’s primary underlying renal pathology influences the survival of both the recipient and the transplanted organ with respect to disease recurrence and associated comorbidities.5
Transplantation candidates undergo extensive immunologic evaluation,8 which helps to avoid transplants that are at risk for antibody-mediated hyperacute rejection. The evaluation consists of four components:
- ABO blood group determination. Determines if the patient is a potential target of recipient-circulating preformed cytotoxic anti-ABO antibody.
- Human leukocyte antigen (HLA) typing (also called tissue typing). This measures the degree of incompatibility between the donor and the recipient. Six HLA antigens are determined for each. (Living donors with a six-antigen match with the recipient allow for decreased intensity of post-transplant immunosuppression.)
- Serum screening for antibodies to HLA phenotypes (PRA score). Sensitization to histocompatibility antigens is of great concern in transplant candidate populations with high levels of HLA antibodies, usually due to having previously received multiple blood transfusions, a prior organ transplant, or pregnancy. These patients will have a high PRA score (the more HLA antibodies, the higher the PRA score), making them more likely to experience immunological issues from a transplant. Transplantation might still be possible, however, if the individual successfully undergoes a desensitizing protocol prior to the surgery.
- Crossmatching. This in-vitro assay method determines whether a potential transplant recipient has pre-formed anti-HLA class I antibodies against the antigens of the kidney donor. A negative crossmatch must be obtained before a kidney is accepted for transplantation.
Transplant patients are also followed up for rejection, immunosuppressant toxicity, and recurrence of kidney disease in the native kidney.
Outcomes – Patient and Graft Survival
Patient and graft survival after kidney transplantation have improved over the past decade. Even so, graft failure is still one of the most common causes of ESKD, accounting for 25% of all patients awaiting renal transplants.5
Life expectancy after renal transplantation depends on patient age, the source of the graft, primary kidney disease, and the presence and degree of comorbidities. Other possible predictive factors include gender, race, and degree of immunosuppression. Data from 2015 provided by the Scientific Registry of Transplant Recipients (SRTR) shows 10-year overall graft survival for both living and deceased donors of approximately 55% to 60% compared with 35% to 40% a decade prior.3 One long-term European study, for example, evaluated determinants of patient survival after renal transplantation among 86 recipients from living donors and 916 from deceased donors. After the first year, an increased risk of death was observed among renal transplant patients >40 years of age, men, patients who received kidneys from deceased donors, patients with diabetes or hypertension, and smokers.3
Early graft loss can be due to vascular thrombosis and rejection, but these occur less often nowadays due to improved surgical techniques and more sophisticated immunosuppressive drugs. Later graft loss is usually due to a combination of factors, including pre-existing donor disease, recurrence of the recipient’s kidney disease, and the recipient’s immunologic response to the new organ. Chronic rejection, another major long-term cause of graft loss, is characterized by slow, progressive renal function deterioration that cannot be altered by common antirejection drugs.
Recurrent Disease
Recurrent renal disease affects as many as 10% of kidney transplant recipients. It accounts for fewer than 3% of all graft losses,5 and is also the third most frequent cause of graft loss at 10 years after transplantation in patients with underlying GN. Although IgAN, the most common type of GN, histologically recurs in up to 60% of kidney transplant patients, only about 5% will lose their graft as a result of its recurrence. Recurrent focal segmental GN and membranoproliferative glomerulonephritis, however, are associated with high risk of graft loss.
ESKD patients with Alport Syndrome receiving renal replacement therapy (RRT) usually have excellent graft survival rates. They also have superior patient survival rates while undergoing dialysis and superior patient and graft survival after transplantation from a deceased donor compared with patients receiving RRT because of other causes of kidney failure. This superior survival might be explained by the lack of additional organ system involvement in Alport Syndrome and the non-recurrent nature of the disease.10 Although individuals with Alport Syndrome are thought to benefit from the non-recurrent character of their disease in their kidney grafts, about 2% to 5% of them will develop anti-glomerular basement membrane (GBM) disease early in the post-transplant period, resulting in rapid graft loss.
Diabetic nephropathy can also recur in renal grafts. Time to onset is similar to that seen in native kidneys. This condition is generally an uncommon cause of graft loss.
Patient and Graft Survival by Source of Graft
The quality of the graft has a direct effect on important clinical outcomes such as acute rejection, delayed graft function, and patient and graft survival. Recipients from related living donors have a lower mortality than recipients from deceased donors, likely because of lower rates of rejection episodes thus less complex immunosuppressive drug regimens.
According to the Scientific Registry of Transplant Recipients, in 2015, the five-year survival for patients who received a deceased-donor kidney in 2010 was 86.8% and for living-donor recipients was 93.5%. Survival was lower in recipients age 65 years and older and in recipients with diabetes as cause of kidney failure.5 Fifteen-year graft failure among adult living donor transplant recipients was 37.3% (1990-2005) and 52.8% for adult deceased donor transplant recipients.11
Expanded-criteria donor (ECD) kidney longevity is believed to be much shorter, with the kidney’s half-life estimated at six to eight years, compared with 10 to 12 years for a non-ECD kidney from a deceased donor.12 ECDs are less-than ideal donors – over age 60 or age 50 to 59 and have two of the following: hypertension; terminal serum creatinine >1.5 mg/dl; or death at any age from cerebrovascular accident.
Patient and Graft Survival by Diagnosis
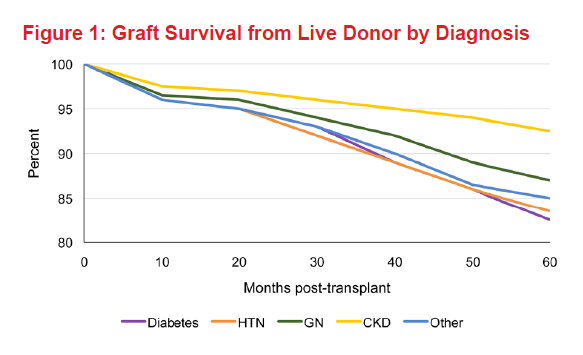
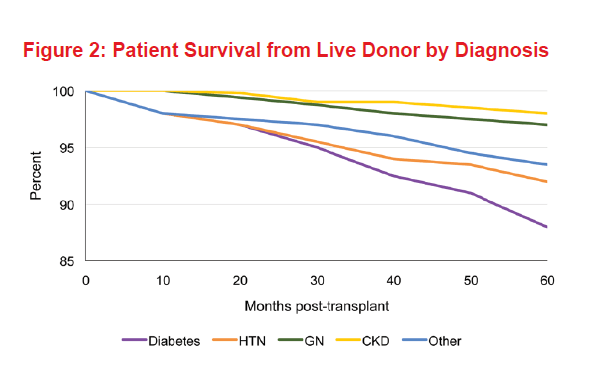
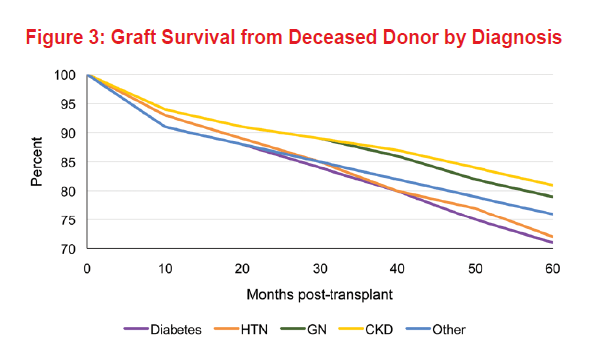
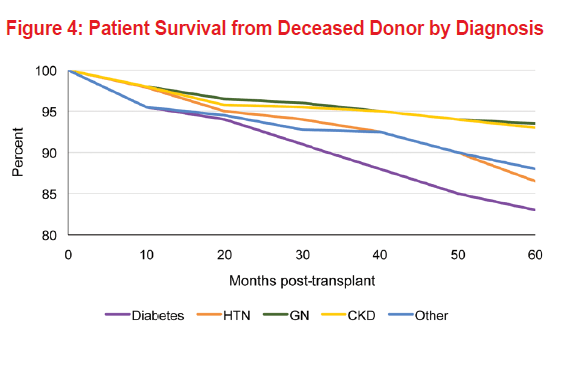
The presence of systemic disorders, particularly vascular disease, is associated with poorer long-term patient survival after renal transplantation. The survival of diabetic patients after renal transplantation is lower than that reported for nondiabetic patients due to the prevalence of extrarenal vascular disease. Five-year graft and patient survival by diagnosis among living donor kidney recipients shows an 83% graft survival and 86.6% patient survival for diabetic subjects (Figures 1 and 2). As expected, for deceased donor kidney recipient diabetic subjects, graft survival was 72% and overall patient survival was 82.5% (Figures 3 and 4).
On the other hand, patients with diseases that primarily affect the kidneys, such as autosomal dominant polycystic kidney disease and GN, have better long-term survival rates post-transplant than those with systemic disorders such as hypertension and diabetes. The same data shows an 87% graft survival and 97% patient survival for living donor kidney recipients with history of glomerular disease, while for deceased donor recipients it was 79% and 92.5%, respectively. Older patients who undergo renal transplantation have higher mortality rates than do younger ones. Among recipients of any age, increased survival is noted with decreased age of the deceased donor, as shown in Figure 4.5
Renal function parameters one year after transplantation are the most important predictors of graft survival. Patient survival at 10 and 20 years has improved and is 75.9% and 64.8%, respectively.1 Ultra-long graft survival (20 years and up) is not uncommon in this day and age. Approximately 25% of kidney transplant patients achieve this rate.
Graft survival after kidney transplantation has improved over the past decade. Death with a functioning graft occurs in about 25% of transplant recipients (15-year data, 1990-2005).11 During the first year after a renal transplant, infections are the leading cause of recipient death. Long-term mortality is more dependent upon other factors, such as coronary artery disease (30.4%), sepsis (27.1%), neoplasm (13%), and stroke (8%).3, 5 The rate of malignancies appears to be directly related to the degree of immunosuppression.
Major morbidities for transplant patients include: hypertension (occurring in 75% to 85% of all recipients); hyperlipidemia (60%); cardiovascular disease (15.8% to 23% – a 10-fold increase over the general population); diabetes (16.9% to 19.9%); osteoporosis (60%); and malignant neoplasms (14%). Post-transplant lymphoma due to Epstein-Barr affects around 2% of recipients. Non-melanoma skin cancer is also particularly high, with human papilloma virus implicated. Diabetes is more likely to be present prior to transplantation, and new-onset diabetes related to post-transplant corticosteroid immunosuppressant drugs.3, 5
Graft and overall survival at 60 months, if hypertension is the primary cause of the ESKD, is 84% and 91.6%, respectively.
Summary and Underwriting Considerations
Insurers need to be aware of these improving outcome trends in renal transplant recipients and refine their underwriting guidelines in accordance with available data on the most important predictors of patient and graft survival.
However, even though long-term kidney graft function has improved, allowing patients with successful kidney transplants to experience enhanced quality and duration of life, insurance companies still need to be aware of the associated comorbidities when they design life, health, and other living benefits insurance products.
Further research in transplant immunology and immunosuppressant agents with the goal of inducing a state of tolerance towards the donor will allow more ultra-long graft survival and further improvements in the morbidity and mortality of kidney transplant recipients.
CASE STUDY, PART I
A 43-year-old male nonsmoker applicant with ESKD received a kidney in 2008 from a living donor. The Panel Reactive Antibody (PRA) score was low. The cause of the ESKD was an unknown primary kidney disease. He is maintaining well on the antirejection drugs tacrolimus and mycophenolate mofetil and no episodes of acute or chronic rejection have occurred. His laboratory evaluations are favorable (normal urinalysis and renal function tests) and he is currently taking an angiotensin-converting enzyme (ACE) inhibitor.
The attending physician report stated that in 2006 the applicant’s primary physician had referred him to a renal clinic as he was complaining of nausea and severe fatigue. At the time, his renal function test results were markedly elevated. Extensive nephrological evaluation did not identify diabetes mellitus or autoimmune diseases such as lupus and vasculitis as underlying causes. Abdominal imaging had revealed small sclerotic kidneys, but no other renal or urinary tract abnormalities. Laboratory evaluation showed bland urinary sediment (i.e., no hematuria, casts, or proteinuria), an estimated glomerular filtration rate (eGFR) of 7 ml/min/1.73m2, and blood pressure of 180/110.
Further medical history questioning revealed that the applicant’s mother had been diagnosed with hematuria in her 30s and died of breast cancer in her late 50s, and a maternal uncle had been diagnosed with ESKD in his 30s and was still alive, on dialysis. In addition, the applicant may have had a few episodes of gross hematuria in his 20s, which he attributed to intense training for a marathon.
The applicant has enjoyed good health until the present time.
Should underwriters be concerned that the applicant’s primary renal disease is unknown (and is this unusual)?
CASE STUDY, PART II
Based on the limited information initially gathered, the applicant was first diagnosed with ESKD due to hypertension, which might have been caused by an undiagnosed glomerular disease. While the exact etiology of his ESKD is unclear, his family history suggested the possibility of hereditary nephritis (i.e., Alport Syndrome).
Hereditary nephritis leading to ESKD is X-linked in about 85% of cases and primarily affects males, is autosomal recessive in 10% to 15% of cases, and autosomal dominant in the remainder. Females tend to have much milder disease. Sensorineural deafness is a characteristic feature observed frequently (but not universally) in patients with Alport Syndrome, and there is no specific treatment.
In the case study, types of GN other than Alport Syndrome should be considered as the possible principal cause. Still, based on the known history, the applicant’s current renal prognosis appears to be more favorable compared to ESKD attributed to diabetic nephropathy or lupus nephritis.
CASE STUDY, PART III
Our applicant is now nine years post-transplant. His graft is functioning well and he has no comorbidities except for history of hypertension, which is managed well with ACEI.
Favorable long-term graft survival predictors, such as young recipient age, living donor HLA matched/low PRA score, early kidney transplant, and optimal first year graft function, make his prognosis quite encouraging.