Thrombocytosis, a condition defined as having a platelet count of more than 450,000 per microliter of blood (450 x 109/L), can be either physiologic in nature or due to primary or secondary causes.
Secondary or reactive thrombocytosis (RT) is far more prevalent than primary or clonal thrombocytosis, and the presence of comorbid conditions in RT, a transient rise in platelets, and lack of genetic mutations favor a secondary etiology. Clinical manifestations of RT can range from no symptoms (most common) to acute thrombosis (rare), and elevated platelet counts may also be a predictor of underlying disease and of mortality. Recent studies of thrombocytosis in patients with COVID-19 infection have shown that elevated platelet counts are predictive of poorer prognosis. Therefore, it is essential for an underwriter to identify RT, as RT is not always a benign entity, which means outcomes can vary.
This article will focus on RT, which is the most common cause of thrombocytosis. It will contrast RT to primary thrombocytosis (PT), outline the factors that favor a diagnosis of RT versus PT, discuss RT’s clinical manifestations and prognosis, and explore RT’s mortality risks and underwriting considerations, particularly in light of the current pandemic.
Introduction
Reactive thrombocytosis (RT), also known as secondary thrombocytosis, is a proliferation of platelets caused by a response to growth factors released from an inflammatory or malignant condition, whereas primary thrombocytosis (PT) is caused by an underlying myeloproliferative or myelodysplastic neoplasm.9
Platelets are the smallest formed elements that circulate in the blood, with a half-life of about four days. They are fragments of larger, multinucleated cells called megakaryocytes, with no nuclei of their own. Platelets are an integral component of coagulation, as proteins on the surface allow platelet adhesion, leading to the formation of a platelet plug.
A normal platelet count ranges from 150,000 to 450,000 platelets per microliter of blood. An abnormally elevated count of >450,000/microL is known as thrombocytosis. Its differential diagnosis is broad. Causes of thrombocytosis can be physiologic (exercise, parturition) and can also be primary/clonal (e.g., essential thrombocythemia, polycythemia vera, primary myelofibrosis, and other hematologic malignancies) as well as secondary/ reactive (due to infectious and inflammatory diseases, neoplasms, anemia, trauma, surgery, asplenia, and iron deficiency).
Thrombocytosis is a common incidental clinical finding, present in 1.5% to 2.2% of the population older than age 40 who are in a primary care setting.4 The incidence of PT (also known as essential thrombocythemia), by comparison, is relatively low, at 1-2.5/100,000 per year.16 Even in extreme thrombocytosis cases, where platelets were greater than 1 million/microL, PT accounted for only 14% of cases. The likelihood of thrombocytosis being due to RT is markedly higher. In fact, a recent study of 777 adults presenting with thrombocytosis revealed that in routine clinical practice, RT accounted for 97% of cases (see Table 1).5
Table 1: Causes of Thrombocytosis |
Condition | Adults (n=777) | Platelet Count of 1000x109/L (n=280) | Children (n=663) |
Infection | 22% | 31% | 31% |
Rebound thrombocytosis | 19% | 3% | 15% |
Tissue damage (surgery, etc.) | 18% | 14% | 15% |
Chronic inflammation | 13% | 9% | 4% |
Malignancy | 6% | 14% | 2% |
Renal disorders | 5% | NS | 4% |
Hemolytic anemia | 4% | NS | 19% |
Postsplenectomy | 2% | 19% | 1% |
Blood loss | NS | 6% | NS |
Primary thrombocytosis | 3% | 14% | 0% |
From Platelets, 4e. Academic Press, 2019.5
RT: Diagnosis of Exclusion
Given the prevalence of RT in clinical practice (and therefore in underwriting), it is important to understand how it is diagnosed. Several factors point toward a reactive etiology rather than a primary cause. The presence of, for example, comorbid conditions such as an infection, a connective tissue disease, bleeding, splenectomy, trauma, or postoperative state would favor a diagnosis of RT, whereas the absence of these conditions would suggest PT.
In adults, acute infection, tissue damage, chronic inflammatory disorders, and malignancy are the most common causes of RT, with one or more of these present in more than 75% of RT cases.6 Transient platelet elevation is also more likely due to RT, whereas concerning signs and symptoms such as vasomotor symptoms, hepatosplenomegaly, and thrombosis tend to be more indicative of PT. If PT is suspected, one would likely see further clinical investigations such as bone marrow biopsy, flow cytometry, and genetic studies.
A growing number of acquired driver mutations causing platelet proliferation through aberrantly activated signaling pathways have been identified, most commonly JAK2V617F.9 Indeed, the latest revision (2016) of the World Health Organization’s diagnostic criteria for essential thrombocythemia (i.e., PT), cited the presence of the JAK2, CALR, or MPL mutation as a criterion, along with a platelet count higher than 450,000/microL and an abnormal bone marrow biopsy. In addition, a minor criterion for PT includes absence of evidence that would result in an RT diagnosis.3
Clinical Setting Determines Risk
RT is typically a clinically silent and transient condition. No clear correlation exists between symptoms and platelet counts; even patients with platelet counts of >1 million/microL due to RT are usually asymptomatic.9
The risk of thrombotic complications from RT is considered to be low, occurring in only 1.6% of patients in one large case series, with all thrombotic events identified as venous and occurring in patients with other risk factors, such as a postoperative state or with underlying malignancy.6
Interestingly, RT in patients with iron deficiency anemia has been associated with a twofold thrombosis risk compared with patients with iron deficiency anemia alone.15 The cause of this association is still not completely understood; however, it is postulated that platelet activation is an ongoing process in iron deficiency and that the frequent comorbidity of iron deficiency with a number of clinical conditions that have known thrombotic risk, such as inflammatory bowel disease and cancer,1 contributes to this phenomenon.
There have rarely been case reports of arterial thrombosis occurring in the setting of RT. In one case report, a 55-year-old female presented with symptoms of left hemiparesis.8 She was diagnosed with a right middle cerebral artery stroke from a right carotid thrombus, which was ultimately determined to be due to RT (>1 million/ microL) secondary to iron deficiency anemia. She was treated with plateletpheresis, a process in which platelets are removed from the bloodstream. Given that the overall risk of thrombosis is considered to be low in RT, treatment with anti-platelet therapy such as aspirin is usually not indicated. Still, treatment can be considered for patients with platelets of >1 million/microL with complications of thrombocytosis, or for patients at risk of developing such complications.12
RT as a Predictor of Poor Outcomes
The presence of RT in infections and cancer may also be predictive of poor outcomes. In a recent study of 421 patients admitted for acute infectious disease, 32 had thrombocytosis with mean peak platelet counts of 527,000/microL. The study authors found that those with thrombocytosis had an increased length of stay, more bacteremia, and increased mortality, with 6.25% of the RT group dying in comparison to 2% of the non-thrombocytosis (control) group.13 This elevated mortality risk has been validated in other investigations on hospitalized patients, with one study showing a 30-day mortality risk 2.5 times higher in those with RT of >500,000/microL compared to those with normal platelet counts.10
Thrombocytosis is a risk marker for cancer. A 2017 prospective study of more than 31,000 patients with thrombocytosis in a primary care setting found that males with thrombocytosis had an 11.6% incidence of cancer in the year following their thrombocytosis diagnosis, and females had an incidence of 6.2%. This compares with 4.1% of males and 2.2% of females who had normal platelet counts. The incidence of cancer rose with age and with higher platelet counts.4
Researchers have also found that an abnormally high platelet count for elderly patients can be an independent predictor of mortality.11 In a study of a large outpatient cohort of asymptomatic elderly individuals, platelet counts and their association with mortality were examined. In a median follow-up period of 3.3 years with a total of 134,132 patient-years of observation, a significant association with increased mortality was found in these patients for both thrombocytosis and thrombocytopenia, with a hazard ratio (HR) of 1.75 for thrombocytosis (see Figure 1). This association persisted across all ethnic groups.
Figure 1:
Overall survival curves stratified by platelet count
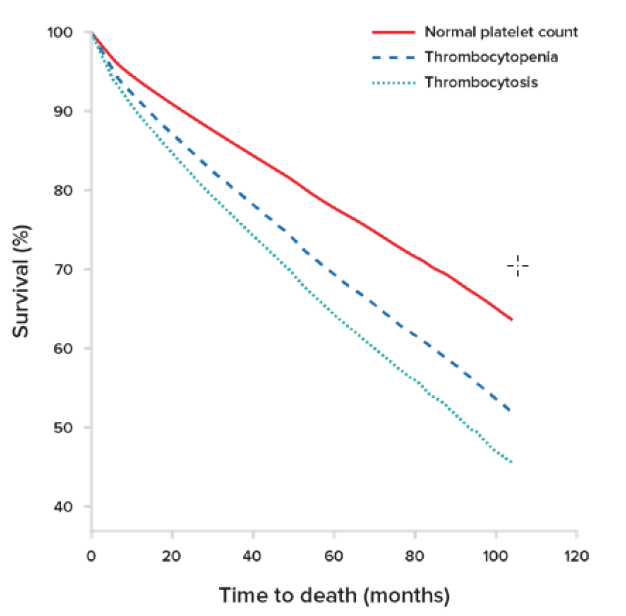
Overall survival curves are stratified by platelet counts after adjustment for all covariates including gender, ethnicity, anemia, neutropenia, and age-adjusted Charison comorbidity index. Both thrombocytopenia and thrombocytosis independently and significantly decreased overall survival compared to that of patients with normal platelet counts (P<0.01).
Adapted from Msaouel, P. Haematologica.11
Thrombocytosis and COVID-19
Significant attention has been paid to thrombosis and COVID-19, as it is thought that elevated platelet counts could be an indicator of cytokine storm, endothelial injury, and/or thrombopoietin release stimulating lung megakaryocytes to produce platelets.2 A literature search of 20 studies with more than 3,500 patients with COVID-19 revealed that higher levels of platelet-to- lymphocyte ratios (PLR), a reflection of a patient’s inflammatory state, were seen in severe COVID-19 disease compared to
non-severe disease.7 This validation of PLR as a prognostic marker in cardiac conditions, tumors, sepsis, pneumonia, and acute respiratory distress syndrome, suggests that RT might also be used as an independent prognostic marker of disease severity in COVID-19.
RT is typically a clinically silent and transient entity… however, it may be predictive of a poor outcome in
acutely and chronically ill individuals.
Reports of central venous sinus thrombosis (CVST) occurring after COVID-19 vaccination has led to further investigation into the hematologic sequelae of the SARS- CoV-2 virus. Remarkably, researchers have identified a novel underlying mechanism for thrombosis in 22 patients presenting with CVST after receiving the ChAdOx1 nCoV- 19 (AstraZeneca) vaccine. These patients did not have thrombocytosis; rather, they were thrombocytopenic. In addition, antibodies to platelet factor 4 were identified in 21 of these patients.14 The authors note, however, the risk of venous thromboembolism after vaccination does not appear to be higher than the background risks in the general population. In fact, a preprint study showed that the incidence of CVST after COVID-19 diagnosis was 10 times higher than CVST incidence after receiving the mRNA vaccine.17 It is evident from these studies that COVID-19 continues to wreak havoc on the hematologic equilibrium of these patients.
Underwriting Considerations
Given what we know about RT, there are several considerations when underwriting thrombocytosis, particularly in light of the ongoing COVID-19 pandemic.
RT is a much more common entity than PT; statistically, a majority of those with thrombocytosis have RT.
It is essential to determine whether a finding of thrombocytosis is reactive or primary.
RT is typically a clinically silent and transient entity with a relatively low thrombotic risk.
RT is associated with higher mortality in acutely ill patients.
RT is an independent predictor of mortality in asymptomatic elderly individuals.
Elevated platelets are associated with worse outcomes in individuals hospitalized with COVID-19.